Global Perspectives on Generics: How Countries Are Cutting Drug Costs and What’s at Stake
When you pick up a prescription, you might not think twice about whether it’s a brand-name pill or a generic. But behind that simple choice lies a complex global system of policies, pricing battles, and regulatory decisions that determine who can afford medicine - and who can’t. From India’s massive generic manufacturing base to South Korea’s strict quality-based pricing tiers, countries are experimenting with wildly different approaches to make drugs cheaper. And while some are saving billions, others are risking shortages, quality issues, and stalled innovation.
How Generics Work - And Why They Matter
Generic drugs aren’t knockoffs. They’re exact copies of brand-name medications, approved after patents expire, with the same active ingredients, dosage, and effectiveness. The key difference? Price. A generic version of a drug can cost 80% less than the original. In the U.S., generics make up 90.1% of all prescriptions filled, but only 23% of total drug spending. That’s because brand-name drugs still carry high prices - even when generics are available.
The real impact shows up in public health systems. The World Health Organization estimates that smart generic policies can cut pharmaceutical spending by 30% to 80%. In Medicare alone, generics saved $142 billion in 2025 - that’s $2,643 per beneficiary. In the European Union, generics account for 65% of all prescriptions but just 22% of costs. These numbers aren’t theoretical. They’re what keeps diabetes, heart disease, and depression treatments accessible to millions.
The U.S. Model: High Volume, High Stakes
The U.S. leads the world in generic use. Why? A mix of strong competition, FDA oversight, and powerful negotiation tools. The FDA’s Orange Book lists over 11,342 approved generic drugs as of December 2024. Companies like Zenara Pharma are getting fast-track approval through the Competitive Generic Therapy (CGT) program, cutting approval time from 18-24 months down to 8-12. Their Sertraline Hydrochloride capsules, approved in August 2025, are a prime example.
But here’s the twist: even though the U.S. has the highest generic usage, its overall drug prices are still among the highest in the world. Why? Because brand-name drugs - especially new ones - remain extremely expensive. Public-sector net prices are 18% lower than in peer countries, but that’s only because generics are so cheap. The real cost burden falls on patients who need newer treatments, and on insurers trying to cover them.
There’s another hidden problem: Pharmacy Benefit Managers (PBMs). These middlemen control which drugs insurance plans cover. Some plans charge higher copays for generics than for brand-name drugs - a bizarre quirk that confuses patients and reduces adherence. Reddit threads from June 2025 show 63% of users frustrated by this. It’s not the drug’s fault. It’s the system.
Europe: Harmonized Rules, Fragmented Prices
Europe has a paradox. The European Medicines Agency (EMA) approves generics for all 27 member states. But once approved, each country sets its own price. That means the exact same pill can cost 300% more in one country than in its neighbor. The OECD’s 2025 report highlights this as a major inefficiency.
Germany uses mandatory generic substitution - pharmacists must switch patients to generics unless the doctor says no. Result? 88.3% of prescriptions are filled with generics. Italy, with similar income levels, only hits 67.4%. Why? No mandatory substitution, weaker education for doctors, and slower reimbursement processes.
Meanwhile, the Netherlands uses external reference pricing. They don’t look at other EU countries. Instead, they compare prices to France, Belgium, the UK, and Norway - and then set their own price lower than all of them. It’s clever. It’s aggressive. And it works: Dutch generic prices are among the lowest in Europe.

China’s Volume-Based Procurement: The Nuclear Option
In 2018, China launched something unprecedented: Volume-Based Procurement (VBP). Instead of letting hospitals choose drugs, the government holds centralized auctions. Manufacturers bid to supply 80% of a hospital’s demand - and the lowest bidder wins.
The results? Average price drops of 54.7%. In some cases, like certain blood pressure meds, prices fell by 93%. By 2025, VBP covered over 400 drugs. But there’s a cost. A 2025 survey by the China Generic Pharmaceutical Association found 23% of manufacturers were selling at a loss. Some stopped production. In 2024, Amlodipine besylate - a common hypertension drug - disappeared from 12 provinces for six to eight weeks. Patients on WeChat forums complained. The government didn’t have enough backup suppliers.
And now, in January 2026, China is expanding VBP to 150 more drugs. Winning bidders must cut prices by 65% on average. Experts warn this could trigger more shortages - and push smaller manufacturers out of business.
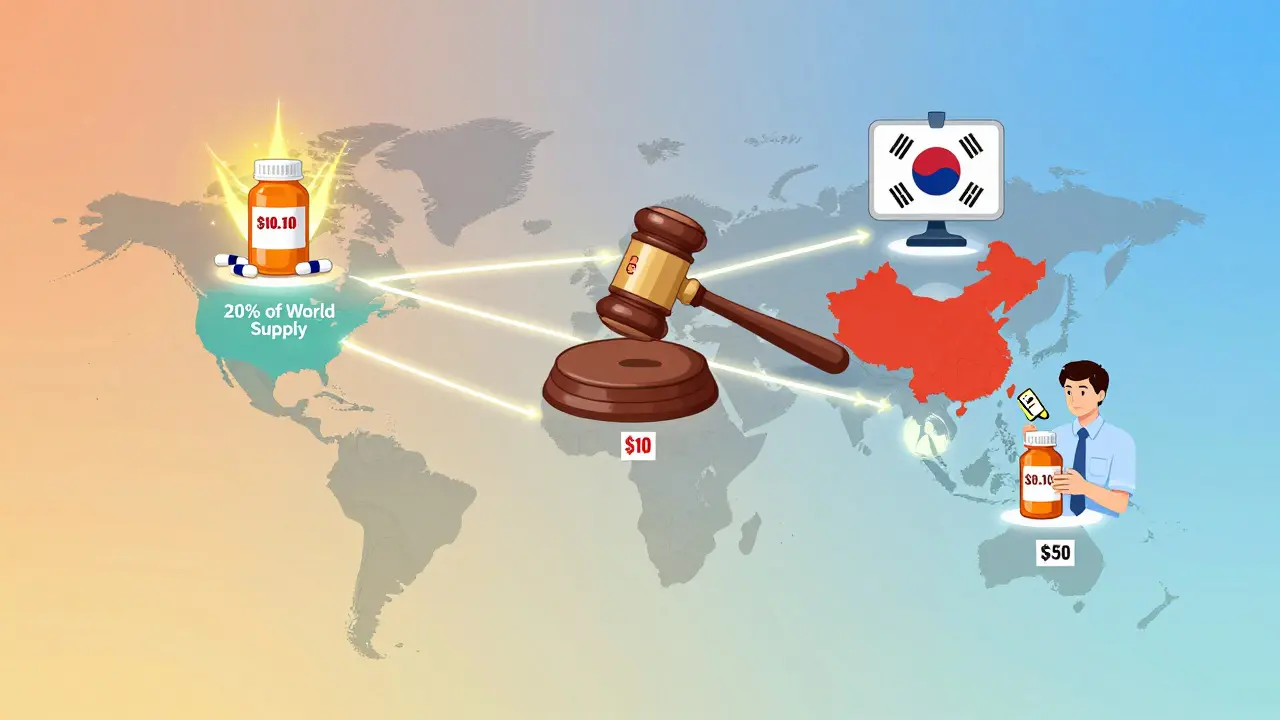
India: The World’s Pharmacy
India produces 20% of the world’s generic drugs by volume. It’s the go-to source for low-cost medicines for Africa, Latin America, and even parts of Europe. How? Section 84 of India’s Patents Act allows compulsory licensing - meaning the government can force a company to let others make a drug, even if it’s still under patent, if it’s too expensive.
This has made India a global powerhouse. But quality is inconsistent. Between 2022 and 2024, the FDA issued 17% more warning letters to Indian manufacturers for data integrity issues - like falsified lab records or skipped tests. Doctors on MedIndia Network report that for critical drugs like antiepileptics or blood thinners, bioavailability varies. One batch might work. The next might not. Patients pay less. But sometimes, they pay with their health.

South Korea: Quality Over Quantity
South Korea took a different path. In 2020, it introduced the “1+3 Bioequivalence Policy.” Only three generic versions of a drug can be approved - and they must all meet strict quality standards. Then, in 2021, it added a pricing system with three tiers:
- 53.55% of brand price - if the generic passes both quality and price tests
- 45.52% - if it passes one
- 38.69% - if it fails both
This cut redundant generic entries by 41% between 2020 and 2024. But it also slashed new generic launches by 29%. Companies say the rules discourage innovation. Why invest in a new generic if you can only be one of three? And why risk quality if the price cap leaves no profit margin?
The Hidden Trade-Off: Affordability vs. Innovation
Every country wants cheaper drugs. But few are talking about the cost of getting them.
When prices are cut too deep - like in China or South Korea - manufacturers stop investing. A 2025 DrugPatentWatch analysis found that aggressive price controls reduced new generic launches by 22-37% compared to pre-reform periods. Fewer generics mean less competition. And less competition means prices rise again - eventually.
There’s also the quality risk. The FDA’s import alerts for quality violations jumped from 1,247 in 2020 to 2,183 in 2024. Most came from countries with weak oversight. The Access to Medicine Foundation warns this could lead to dangerous failures - especially for drugs with narrow therapeutic windows, like warfarin or lithium.
Meanwhile, the U.S. and EU are watching the clock. Between 2025 and 2030, $217-236 billion in annual branded drug sales will lose patent protection. If policies don’t adapt, supply chains will collapse under the weight of demand. The WHO says sustainable pricing - with manufacturers earning at least 15-20% gross margin - is the only way to ensure consistent quality and supply.
What’s Next? Three Big Trends
Three forces will shape the next decade:
- The U.S. Inflation Reduction Act: Starting in 2028, Medicare will negotiate prices for 10-20 high-cost drugs annually. This could cut originator revenues by 25-35% and push more patients toward generics.
- EU Pharmaceutical Package: Expected in late 2025, this aims to harmonize pricing and speed up generic entry. If passed, it could cut approval delays by 12-15% across Europe.
- Consolidation: McKinsey predicts the number of global generic manufacturers will drop from 3,500 to 2,200 by 2030. Only companies with full control over R&D, manufacturing, and distribution will survive.
The future of generics isn’t just about cost. It’s about trust. Can we make drugs affordable without sacrificing safety? Can we reward innovation while keeping medicines within reach? Right now, no country has cracked it perfectly. But the lessons from the U.S., China, India, and South Korea are clear: balance matters. And without it, the cheapest drug in the world becomes useless if no one can get it.
Are generic drugs as effective as brand-name drugs?
Yes. By law, generics must contain the same active ingredient, strength, dosage form, and route of administration as the brand-name version. They must also meet strict bioequivalence standards - meaning they deliver the same amount of drug into the bloodstream at the same rate. The FDA requires generics to be within 80-125% of the brand’s absorption levels. Millions of patients use generics safely every day. Studies show no meaningful difference in outcomes for most conditions.
Why do some people say generics don’t work as well?
This usually comes from two sources. First, for drugs with a narrow therapeutic index - like warfarin, levothyroxine, or certain seizure meds - tiny changes in absorption can matter. Even if a generic meets FDA standards, some patients feel different switching brands. Second, poor-quality generics from unregulated manufacturers - especially in emerging markets - sometimes fail to meet standards. The FDA has seen a 75% increase in import alerts since 2020, mostly from plants with data integrity issues. It’s not the generic model that’s flawed - it’s the enforcement.
Why are generic prices so different between countries?
Because drug pricing isn’t regulated globally. Each country decides how much to pay. The U.S. uses market competition and Medicare negotiation. Germany uses mandatory substitution and reference pricing. China uses bulk auctions. India uses compulsory licensing. The Netherlands picks the lowest price from non-EU countries. There’s no universal rule - just a patchwork of local policies, negotiation power, and political pressure. That’s why the same pill can cost $0.10 in India and $3.50 in the U.S.
Can generic drug shortages be prevented?
Yes - but it requires planning. Shortages happen when manufacturers can’t make a profit. In China, VBP price cuts led to 23% of firms operating at a loss. In South Korea, strict limits on how many generics can be approved reduced competition. The solution? Maintain reasonable margins (15-20% is recommended by WHO), diversify supply chains, and require manufacturers to report production capacity. Countries that keep backup suppliers and monitor inventory levels - like the U.S. and Germany - rarely face major shortages.
What’s the biggest threat to the future of generics?
The biggest threat isn’t patents - it’s quality erosion. As price pressure grows, some manufacturers cut corners on testing, clean rooms, and raw materials. The FDA’s rise in import alerts proves this is already happening. If patients start losing trust in generics because of unsafe products, they’ll return to expensive brands - and healthcare costs will spike again. Sustainable affordability requires strong oversight, not just low prices.




Mariah Carle
So we’re basically playing Jenga with people’s lives, huh? 🤔 One wrong move - like cutting prices too deep in China - and the whole tower collapses. Generics are supposed to save us, not turn into a game of Russian roulette with your meds. I just hope someone’s keeping track of who’s dying because a $0.10 pill didn’t dissolve right. 😔
Justin Rodriguez
The FDA’s import alert numbers are terrifying. 2,183 in 2024 vs. 1,247 in 2020? That’s not just a trend - that’s a systemic failure. Most of these violations are from facilities with poor QA systems, often because manufacturers are forced to operate on razor-thin margins. It’s not greed. It’s survival. And when survival means cutting corners on clean rooms or skipping stability tests, we all lose. We need margins that allow quality, not just low prices.
Raman Kapri
India is not the world’s pharmacy. India is the world’s pharmacy *problem*. Yes, we make 20% of the world’s generics - but 17% more FDA warning letters since 2022? That’s not a badge of honor. That’s a red flag. You can’t export quality when your domestic regulatory oversight is a joke. And don’t even get me started on compulsory licensing - it’s economic vandalism disguised as social justice. The world needs generics. But not *this* kind.
Megan Nayak
Let’s be real: the U.S. doesn’t have low drug prices because of generics. We have low generic prices because we have 90% market penetration - but the *real* prices? Still insane. PBMs are the real villains here. They charge higher copays for generics than brands? Are you kidding me? That’s not a bug - that’s a feature of a system designed to confuse and gouge. Meanwhile, the WHO says manufacturers need 15-20% margins? Yeah, right. The same people who profit off $500 insulin pills are now crying about ‘sustainability.’ Please. 😂
Tildi Fletes
South Korea’s 1+3 Bioequivalence Policy is one of the most thoughtful frameworks I’ve seen. By limiting competition to only the highest-quality generics and tiering pricing based on performance, they incentivize innovation *within* the generic space. It’s not about quantity - it’s about reliability. And yes, it reduces new entrants. But that’s not a flaw. It’s a filter. Too many generics = too many risks. Quality control should never be sacrificed for volume. This model deserves global study.
Siri Elena
Oh look, another ‘thought-provoking’ article that treats generics like a moral dilemma instead of a supply chain problem. Let me guess - the solution is ‘balance’? Wow. Revolutionary. Next you’ll tell me sunshine is good for plants. Meanwhile, in the real world, someone’s kid is missing their seizure med because China’s auction system drove a manufacturer into bankruptcy. And we’re having a philosophical debate about ‘trust.’ Cute.
Levi Viloria
I’ve been on generic meds for 8 years. Never had an issue. But I know people who swear their ‘new’ generic made them dizzy or gave them headaches. Turns out, for drugs like levothyroxine or warfarin, even tiny bioavailability shifts matter. Not because generics are bad - because the system doesn’t account for individual sensitivity. We need better tracking. Not more generics. More awareness.
Zacharia Reda
I work in pharma logistics. Let me tell you something no one’s saying: the biggest threat to generics isn’t price - it’s *consolidation*. By 2030, we’ll have 2,200 manufacturers left from 3,500. That means fewer backup suppliers. One factory fire, one FDA shutdown, one failed audit - and suddenly 12 provinces in China are out of amlodipine. We’re building a fragile system on top of a fragile supply chain. And we’re pretending it’s sustainable. It’s not.
Matt Alexander
Simple truth: if a drug costs less than a coffee, someone’s getting screwed. Either the maker, the worker, or the patient. You can’t have cheap, safe, and reliable without paying for it somewhere. Stop pretending there’s a magic button.