Hemolytic Anemia from Medications: Recognizing Red Blood Cell Destruction
Hemolytic Anemia Risk Assessment Tool
This tool helps identify potential risk factors for drug-induced hemolytic anemia based on medications taken, symptoms, and G6PD status. Not a medical diagnosis.
When a medication triggers your body to destroy its own red blood cells, it’s not just a side effect-it’s a medical emergency. Drug-induced immune hemolytic anemia (DIIHA) is rare, but when it happens, it can drop your hemoglobin by 3 to 5 grams per deciliter in under 72 hours. That’s like losing a full pint of blood overnight. And because the symptoms start slow-fatigue, pale skin, a racing heart-it’s easy to miss. By the time jaundice shows up, your liver is already struggling to process the flood of broken-down cells. This isn’t theoretical. It’s happening in hospitals right now, often because the link between the drug and the crash in red blood cells wasn’t recognized in time.
How Medications Turn Against Your Blood Cells
Not all drug-induced hemolysis works the same way. There are two main paths, and knowing which one you’re dealing with changes everything.
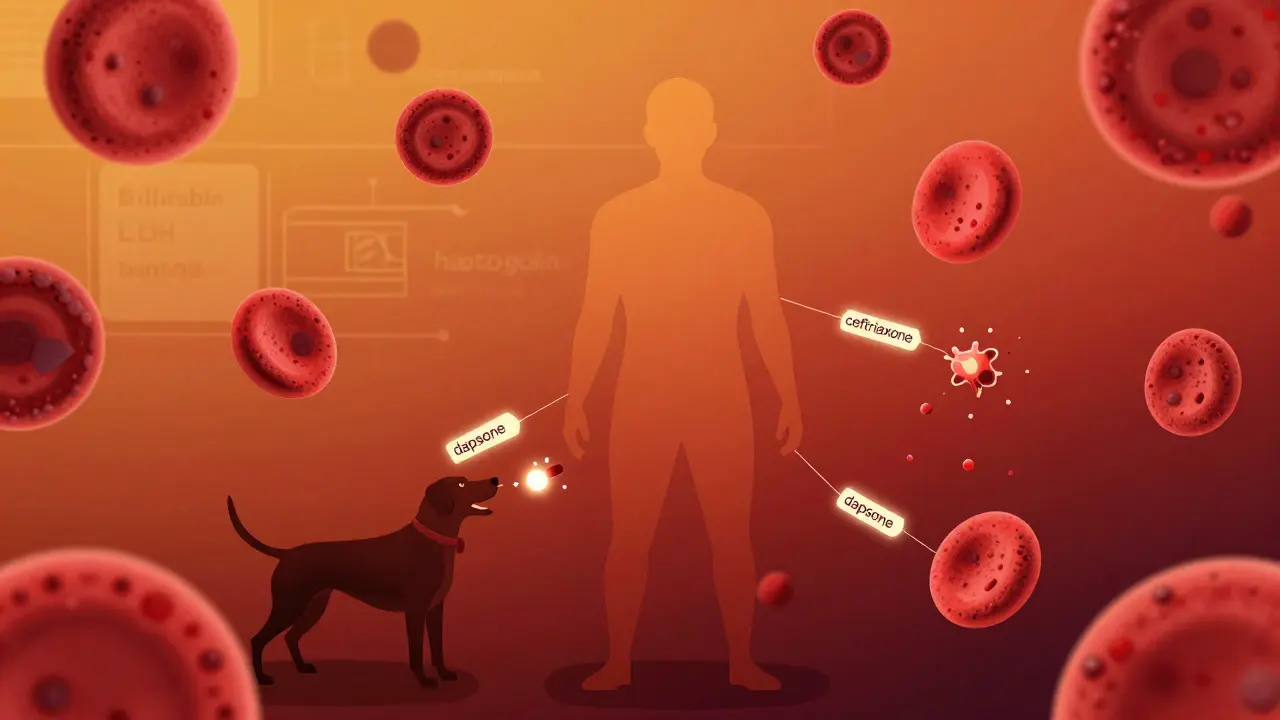
The first is immune-mediated. Here, the drug binds to your red blood cells like a sticky tag. Your immune system sees that tag as foreign, sends out antibodies, and those antibodies latch onto the cells like hunters marking prey. Then your spleen starts pulling them apart. The most common culprits? Cephalosporins. Specifically, cefotetan, ceftriaxone, and piperacillin. Together, they cause about 70% of all immune cases. Methyldopa used to be a big one, but fewer people take it now. Still, if someone’s on a third-generation cephalosporin for a stubborn infection and suddenly starts looking pale and tired, this needs to be on the radar.
The second path is oxidative. This one doesn’t involve antibodies. Instead, the drug directly attacks hemoglobin inside the red blood cell, turning it into a clumpy, useless mess called Heinz bodies. These clumps rip holes in the cell membrane, and the cell bursts. This happens fast-sometimes within 24 hours. The big risk factor? G6PD deficiency. It’s an inherited condition, common in African American men (10-14%) and people of Mediterranean descent (4-15%). But even if you don’t know you have it, certain drugs can still trigger this. Dapsone, phenazopyridine (Pyridium), ribavirin, and even topical benzocaine from throat sprays or dental gels can do it. The scary part? You might never have had symptoms before. One sip of a medication you’ve taken before, and suddenly your body is destroying red cells like there’s no tomorrow.
What You’ll Feel-and What the Lab Shows
Symptoms are vague at first. Fatigue? Yes. Weakness? Of course. But if you’re also noticing your skin turning yellow, your urine darkening like cola, or your heart pounding even when you’re sitting still, that’s not just being tired. That’s your body screaming.
Here’s what the numbers look like in confirmed cases:
- 92% report fatigue
- 87% feel weak
- 76% get short of breath
- 73% have pale skin
- 81% show jaundice
- 68% have a heart rate over 100 bpm
But symptoms alone won’t confirm it. Lab tests are the real key. The classic triad:
- Indirect bilirubin >3 mg/dL
- Lactate dehydrogenase (LDH) >250 U/L
- Haptoglobin <25 mg/dL
And the blood smear? That’s where you’ll see the proof. Spherocytes-small, round, dense red cells-mean immune destruction. Heinz bodies-dark clumps clinging to the cell edges-mean oxidative damage. If you’re seeing either, you’re looking at hemolytic anemia.
One big trap: the direct antiglobulin test (DAT). It’s positive in 95% of immune cases. But if you test too early, or if the drug is acting in a weird way, it can be negative. That doesn’t rule it out. You still have to look at the whole picture.
The Drugs That Cause the Most Trouble
There are over 100 medications linked to oxidative hemolysis. But only a handful are common enough to be worth memorizing.
Immune-mediated triggers (most common):
- Cephalosporins (cefotetan, ceftriaxone, piperacillin)
- Methyldopa
- Penicillin derivatives
- NSAIDs (like ibuprofen or naproxen in rare cases)
- Nitrofurantoin
- Levofloxacin
- Dapsone
Oxidative triggers (dangerous in G6PD deficiency or alone):
- Dapsone
- Phenazopyridine (Pyridium)
- Ribavirin
- Primaquine
- Sulfa drugs
- Topical benzocaine
- Amyl nitrate, butyl nitrate
Notice how some drugs appear on both lists? Dapsone can trigger both immune and oxidative reactions. That’s why a full drug history matters-not just what’s new, but what’s been taken long-term.
What Happens When You Don’t Stop the Drug
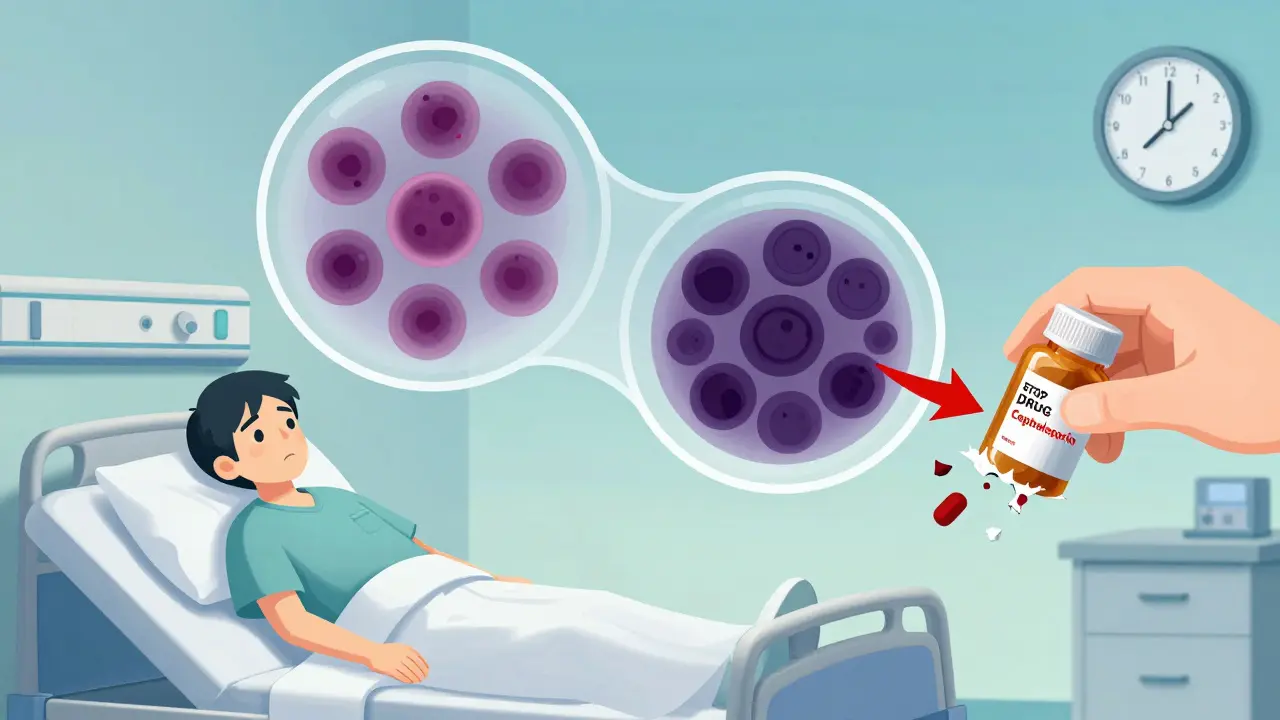
Continuing the drug? That’s how you go from bad to critical.
Severe cases can drop hemoglobin below 6 g/dL. At that level, your heart is working overtime just to pump enough oxygen. Studies show 22% develop arrhythmias, 15% get heart muscle damage, and 8% end up in heart failure. That’s not a stretch-it’s a direct result of the body being starved of oxygen because its red cells are being destroyed faster than they can be replaced.
And here’s another hidden danger: clotting. People with DIIHA are often hypercoagulable. One 2023 study found 34% of severe cases developed venous clots-deep vein thrombosis or pulmonary embolism. So even though you’re losing blood, you’re also at risk of forming dangerous clots. It’s counterintuitive, but real.
How to Treat It-And What Not to Do
Step one is simple: stop the drug. Immediately. No waiting. No “let’s see if it gets better.” If you suspect DIIHA, the drug is guilty until proven innocent. And in most cases, it is.
After stopping the drug:
- Most people stabilize within 7-10 days.
- Full recovery usually takes 4-6 weeks.
Transfusions? Only if hemoglobin is below 7-8 g/dL or symptoms are severe. Don’t transfuse just because the number looks low. You’re giving someone healthy cells into a system that’s still attacking them. You need to break the cycle first.
Corticosteroids? They’ve been used for decades. But studies show they don’t change outcomes much. The body often heals itself once the drug is gone. So use them only if the immune response is raging and not slowing down.
For stubborn cases-where antibodies keep circulating even after stopping the drug-treatment escalates. IV immunoglobulins (1 g/kg/day for two days) can block the immune attack. If that fails, rituximab (a monoclonal antibody that wipes out B cells) works in 78% of refractory cases within 3-6 weeks.
Now, the critical warning: methylene blue. It’s used to treat severe methemoglobinemia (when iron in hemoglobin gets stuck in the wrong state). But if the patient has G6PD deficiency, methylene blue can trigger massive hemolysis. It’s a death sentence in those cases. Always check G6PD status before giving it.
And here’s a sneaky pitfall: G6PD testing during active hemolysis. The test measures enzyme levels in older red cells. But during an attack, those old cells are gone. What’s left are new ones with normal enzyme levels. So you get a false negative. Wait 2-3 months after recovery to test properly. Otherwise, you’ll miss the diagnosis-and put the person at risk again.

Why This Is Still Missed in Hospitals
A 2024 study of internal medicine residents found they correctly diagnosed DIIHA only 58% of the time on first try. After targeted training? That jumped to 89%. Why? Because most doctors don’t think of it. Fatigue? Probably anemia from iron deficiency. Jaundice? Liver issue. Rapid heartbeat? Stress. It’s not until someone connects the dots-medication, timing, lab pattern-that the light turns on.
One hospital in Manchester started using EHR alerts. If a patient was prescribed ceftriaxone and their bilirubin or LDH spiked within 10 days, the system flagged it. Over 18 months, severe DIIHA cases dropped by 32%. That’s not luck. That’s system design.
Patients don’t always remember every drug they’ve taken. That’s why pharmacists need to be part of the team. A simple question: “Have you taken any new antibiotics, painkillers, or urinary tract meds in the last 2 weeks?” can save a life.
What You Need to Remember
- Drug-induced hemolytic anemia is rare-but deadly if missed.
- Two mechanisms: immune (antibodies) and oxidative (direct damage).
- Cephalosporins are the #1 immune trigger; dapsone and phenazopyridine are top oxidative triggers.
- Stop the drug immediately. That’s the single most important step.
- Don’t rely on G6PD testing during active hemolysis-wait 2-3 months.
- Methylene blue can kill G6PD-deficient patients-never give it without confirming status.
- Lab triad: high bilirubin, high LDH, low haptoglobin.
- Transfusions are for severe symptoms, not just low numbers.
If you’re on a medication and suddenly feel worse-especially with jaundice, dark urine, or heart palpitations-don’t wait. Ask: could this be my drugs? Because sometimes, the cure isn’t more medicine. It’s stopping the one you’re already taking.
Can a common painkiller like ibuprofen cause hemolytic anemia?
Yes, but it’s rare. NSAIDs like ibuprofen and naproxen have been linked to immune-mediated hemolytic anemia in isolated cases. Most often, it happens after long-term use, and the reaction is unpredictable. It’s not a common side effect, but if you’ve been taking NSAIDs daily for months and suddenly develop unexplained fatigue or jaundice, it’s worth checking.
Is hemolytic anemia from drugs more dangerous in older adults?
It can be. Older adults often have reduced kidney or liver function, which slows drug clearance. They’re also more likely to be on multiple medications, increasing the chance of interaction. Plus, their hearts are less able to handle sudden drops in oxygen. A hemoglobin drop that might be manageable in a 30-year-old can trigger heart failure in someone over 65. Age isn’t the direct cause, but it increases risk.
If I have G6PD deficiency, what medications should I avoid?
Avoid dapsone, phenazopyridine (Pyridium), primaquine, sulfonamides, ribavirin, and methylene blue. Even topical benzocaine in throat sprays or dental gels can trigger hemolysis. Always check with your pharmacist before starting any new medication-prescription, OTC, or herbal. Keep a list of unsafe drugs handy. Many clinics now provide wallet cards for G6PD-deficient patients.
How long does it take to recover after stopping the drug?
Most people start to recover within 7-10 days after stopping the drug. Hemoglobin levels usually stabilize by then. Full recovery-meaning red blood cell production catches up and symptoms disappear-takes about 4 to 6 weeks. The body regenerates red cells naturally, but it needs time. Don’t rush back to normal activity too soon. Fatigue can linger.
Can children get drug-induced hemolytic anemia?
It’s very rare in children, but it does happen. When it does, it often presents more severely than in adults. One study found children had average hemoglobin levels of 5.2 g/dL at diagnosis, compared to 6.8 g/dL in adults. That means they’re more likely to need transfusions. Always consider medication history-even a single dose of an antibiotic or fever reducer can trigger it in susceptible kids.
Is there a way to prevent this before it starts?
Yes, in some cases. If you know you have G6PD deficiency, avoid known oxidative triggers. Ask your doctor to screen for high-risk drugs before prescribing. Hospitals are starting to use electronic alerts that flag dangerous combinations. If you’ve had a previous episode of drug-induced hemolysis, keep a list of the culprit drugs and share it with every provider you see. Prevention is about awareness, not just testing.


Adam M
Cephalosporins are the usual suspects. If someone's on ceftriaxone and drops hemoglobin 4 g/dL in 48 hours? Stop the drug. Now. No waiting for labs.
Richard Harris
this is so important i cant believe more docs dont talk about this. i had a patient last month who looked fine until she turned yellow. scary stuff.
Noluthando Devour Mamabolo
G6PD deficiency is a silent time bomb 🚨 Especially in populations with high prevalence. We need better screening protocols before prescribing oxidant drugs. #HemolysisAwareness
Stephanie Paluch
i just read this and my heart sank. my aunt had this happen after a dental procedure. they didn't connect it until she was in the ICU. please, if you're on benzocaine sprays and feel off-listen to your body 💔
Kandace Bennett
Americans really need to stop treating meds like candy. This is why we have a healthcare crisis. If you're taking ceftriaxone for a sinus infection, you're doing it wrong. 🇺🇸
Tim Schulz
Oh wow. So the drug that's supposed to kill the infection... is now killing your RBCs? 🤡 Classic pharma magic. Next they'll say aspirin 'accidentally' dissolves your kidneys.
Jinesh Jain
Interesting read. I've seen dapsone cause hemolysis in leprosy patients. The key is early detection. Labs don't lie.
Ali Hughey
This is a BIG PHARMA cover-up. They know cephalosporins trigger hemolysis. They bury the data. They profit from transfusions. The FDA is complicit. Wake up people. This is systematic. 🕵️♂️💉
Alex MC
This is exactly why we need better pharmacovigilance. Not every reaction is rare. Some are just underreported. We owe it to patients to track these patterns harder.
rakesh sabharwal
The fact that this isn't in every med school curriculum is criminal. You can't be a competent clinician without understanding drug-induced hemolysis. This is basic. Pathophysiology 101.
Aaron Leib
I’ve had patients bounce back fast once we stopped the drug. Early recognition saves lives. Keep pushing this awareness. You’re doing great work.
Dylan Patrick
G6PD deficiency is one of those things you never think about… until your kid’s urine turns brown after a fever med. Then you Google. And panic. We need better patient education. Period.
Kathy Leslie
My grandma died from this. No one ever told us the antibiotic could do this. Please, if you're reading this-ask your doctor about G6PD before taking anything new. It's simple. It matters.
Adam M
And if you're on methyldopa? Still a risk. Even if it's old school. Don't assume it's safe.